 
All salts of sulfate are soluble except for barium sulfate, lead(II) sulfate, and strontium sulfate. An alkaline earth metal, strontium is a soft silver-white yellowish metallic element. chromium(III) nitrate (aq) + iron(II) sulfate (aq). Referring to the solubility rules, MgCO 3 is insoluble in water resulting in a phase label of ( s), while NH 4Br is soluble in water resulting in a phase label of ( aq).\) which is added to municipal water supplies to kill microorganisms. Strontium is a chemical element it has symbol Sr and atomic number 38. Ammonium ions pair up with bromide ions forming NH 4Br (NH 4 + balances the charge of Br –). Comments (0) Write the balanced molecular and net ionic equations for each of the following neutralization reactions: (a) Aqueous acetic acid is neutralized by aqueous barium hydroxide. Magnesium ions pair up with carbonate ions forming magnesium carbonate, MgCO 3 (Mg 2 + balances the charge of CO 3 2 –). Aqueous solutions of barium chloride and lithium sulfate are mixed. All salts of sulfate are soluble except for barium sulfate, lead(II) sulfate, and strontium sulfate. Write the net ionic equation for any reaction that occurs. MgBr 2 is called magnesium bromide and is comprised of magnesium ions, Mg 2 +, and bromide ions, Br −, while (NH 4) 2CO 3 is called ammonium carbonate and is comprised of ammonium ions, NH 4 +, and carbonate ions, CO 3 2 –. chromium(III)nitrate(aq) + iron(II)sulfate(aq). sulfate and strontium iodide chromium (III mmoTi5jcadelenan mmoTi5jcadelenan Chemistry High School answered expert verified Write a molecular. If no reaction occurs, leave all boxes blank and click on Submit.) a. (Be sure to specify states such as (aq) or (s). (OpenStax, CC BY 4.0, via Wikimedia Commons)Ĭomplete and balance the following equation. If no reaction takes place, write "No Reaction" after the arrow. Question: Write net ionic equations for the reaction, if any, that occurs when aqueous solutions of the following are mixed. HCl(aq) + LiOH(aq) rightarrow H2O(l) + LiCl(aq) b. a net ionic equation is written, omitting the spectator ions. The complete chemical equation can be written to describe what happens, and such an equation is useful in making chemical. chromium(III) nitrate and sodium phosphate Write balanced complete ionic and net ionic equations for each reaction. A typical precipitation reaction occurs when an aqueous solution of barium chloride is mixed with one containing sodium sulfate. potassium sulfate and strontium iodide c. 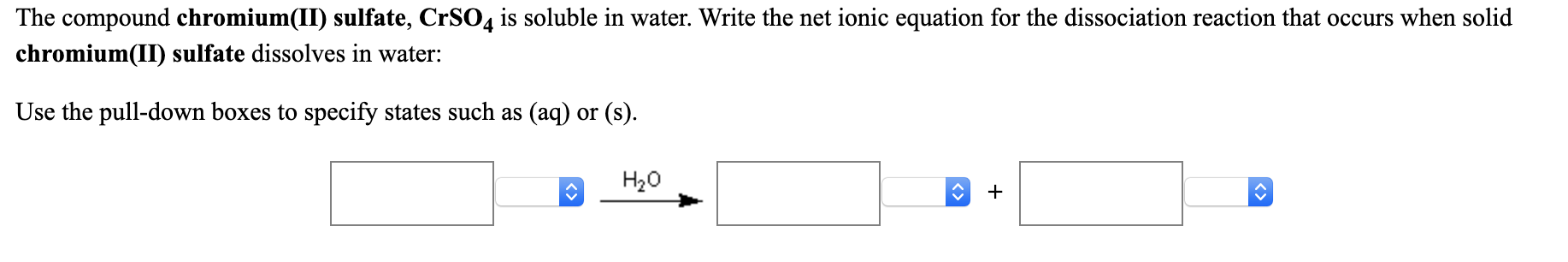
If no reaction occurs, leave all boxes blank and click on Submit a chromium (III) chloride and sodium hydroxide. If no reaction occurs, write 'NO REACTION.' a. \): An x-ray of the digestive organs of a patient who has swallowed a “barium milkshake.” A barium milkshake is a suspension of very fine BaSO 4 particles in water the high atomic mass of barium makes it opaque to x-rays. Write net ionic equations for the reaction, if any, that occurs when aqueous solutions of the following are mixed (Be sure to specify states such as (aq) or (s). 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
